Unit 3 covers energy in reactions, cellular respiration, and photosynthesis. Different types of reactions increase or decrease free energy. Cellular respiration reduces free energy by converting chemical energy to ATP through complex system interactions. Photosynthesis similarly reduces free energy by converting light energy to chemical energy through system interactions. These different ways of converting energy, in mitochondria and chloroplast, are a result of evolution.

1. List examples of endergonic and exergonic reactions
- Endergonic reactions absorb energy in order to happen such as dissolving salt in water and photosynthesis.
- Exergonic reactions release energy such as glycolysis and the breakdown of ATP to ADP.
(1)RSC [http://www.rsc.org/Education/Teachers/Resources/cfb/metabolism.htm]
(2)Meta[http://metabolismcook.wikispaces.com/Exergonic+and+Endergonic+Reactions]
- Endergonic reactions absorb energy in order to happen such as dissolving salt in water and photosynthesis.
- Exergonic reactions release energy such as glycolysis and the breakdown of ATP to ADP.
(1)RSC [http://www.rsc.org/Education/Teachers/Resources/cfb/metabolism.htm]
(2)Meta[http://metabolismcook.wikispaces.com/Exergonic+and+Endergonic+Reactions]
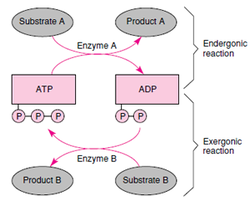
2. Determine the key role of ATP in energy coupling
- ATP powers cellular work by coupling exergonic and endergonic reactions. In the exergonic reaction, ATP release hydrolysis of ATP to release phosphate group through hydrolysis. The released energy of ATP is coupled with endergonic processes by transferring a phosphate group to another molecule.
(1)UIC[http://www.uic.edu/classes/bios/bios100/lecturesf04am/lect04.htm]
(2)TRU[http://www.trueorigin.org/atp.asp]
- ATP powers cellular work by coupling exergonic and endergonic reactions. In the exergonic reaction, ATP release hydrolysis of ATP to release phosphate group through hydrolysis. The released energy of ATP is coupled with endergonic processes by transferring a phosphate group to another molecule.
(1)UIC[http://www.uic.edu/classes/bios/bios100/lecturesf04am/lect04.htm]
(2)TRU[http://www.trueorigin.org/atp.asp]
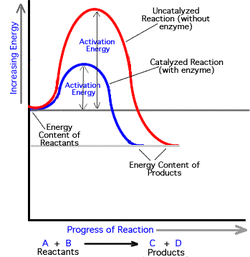
3. Recognize that enzymes work by lowering the energy of activation
The energy of activation is the the amount of energy it takes to start a reaction. Enzymes are proteins serve as catalysts, because enzymes can speed up the reactions by lowing the energy of activation.
(1) AUS[http://www.austincc.edu/~emeyerth/enzymes.htm]
(2) ESY[http://www2.estrellamountain.edu/faculty/farabee/biobk/biobookenzym.html]
The energy of activation is the the amount of energy it takes to start a reaction. Enzymes are proteins serve as catalysts, because enzymes can speed up the reactions by lowing the energy of activation.
(1) AUS[http://www.austincc.edu/~emeyerth/enzymes.htm]
(2) ESY[http://www2.estrellamountain.edu/faculty/farabee/biobk/biobookenzym.html]
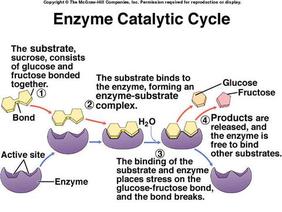
4. llustrate the catalytic cycle of an enzyme that results in the production of a final product
The substrate bind to the active site of an enzyme and form a complex called an enzyme-substrate complex. Then the substrate is converted to product and the product will be released from the enzyme.
(1) Uci[http://www.uic.edu/classes/bios/bios100/lecturesf04am/lect04.htm]
(2) Ss[http://www.slideshare.net/mgsonline/catalysis]
The substrate bind to the active site of an enzyme and form a complex called an enzyme-substrate complex. Then the substrate is converted to product and the product will be released from the enzyme.
(1) Uci[http://www.uic.edu/classes/bios/bios100/lecturesf04am/lect04.htm]
(2) Ss[http://www.slideshare.net/mgsonline/catalysis]

5. Identify factors that influence enzyme activity
- pH: The enzyme usually work at pH of 7. If the pH is too high, the enzyme is denatured and doesn't function anymore.
- Temperature: Enzymes usually work at the temperature of 37 degree. The higher temperature can faster the enzyme work because the substrates are moving faster. However, if the temperature is too high or low, the enzyme will be denatured and not function anymore.
- Inhibitors: Inhibitors can stop enzyme functioning. One is competitive inhibitors is compete with substrate for the active site of enzyme. The other is noncompetitive inhibitors that bind to another site of enzyme to cause the enzyme change shape, so the substrate cannot bind with the enzyme.
- Substrate concentration: As the substrate concentration increases, the reaction rate increases. At higher concentrations the enzyme molecules become saturated and the rate will be constant
- Enzyme concentration: As the enzyme concentration increases, the reaction rate increases because there are more enzyme can work. The rate will be constant when the enzyme become saturated.
(1) ALe [http://alevelnotes.com/Factors-affecting-Enzyme-Activity/146?tree=]
(2) Buzzle [http://www.buzzle.com/articles/important-factors-that-influence-enzyme-activity.html]
- pH: The enzyme usually work at pH of 7. If the pH is too high, the enzyme is denatured and doesn't function anymore.
- Temperature: Enzymes usually work at the temperature of 37 degree. The higher temperature can faster the enzyme work because the substrates are moving faster. However, if the temperature is too high or low, the enzyme will be denatured and not function anymore.
- Inhibitors: Inhibitors can stop enzyme functioning. One is competitive inhibitors is compete with substrate for the active site of enzyme. The other is noncompetitive inhibitors that bind to another site of enzyme to cause the enzyme change shape, so the substrate cannot bind with the enzyme.
- Substrate concentration: As the substrate concentration increases, the reaction rate increases. At higher concentrations the enzyme molecules become saturated and the rate will be constant
- Enzyme concentration: As the enzyme concentration increases, the reaction rate increases because there are more enzyme can work. The rate will be constant when the enzyme become saturated.
(1) ALe [http://alevelnotes.com/Factors-affecting-Enzyme-Activity/146?tree=]
(2) Buzzle [http://www.buzzle.com/articles/important-factors-that-influence-enzyme-activity.html]

6. Write out the summary equation of cellular respiration
- C6H12O6+6O2→ 6CO2 + 6H2O + Energy ( ATP+heat)
(1) WYZ [http://www.wyzant.com/resources/lessons/science/biology/cellular-respiration]
(2) Lab [ http://faculty.southwest.tn.edu/rburkett/GB%201%20cell%20resp.htm]
- C6H12O6+6O2→ 6CO2 + 6H2O + Energy ( ATP+heat)
(1) WYZ [http://www.wyzant.com/resources/lessons/science/biology/cellular-respiration]
(2) Lab [ http://faculty.southwest.tn.edu/rburkett/GB%201%20cell%20resp.htm]

7. Differentiate between fermentation and cellular respiration
- Cellular respiration is an aerobic reaction which requires oxygen. Fermentation does not require oxygen but does release lactic acid which causes our cells and muscles to weaken.
(1) Clc [http://biology.clc.uc.edu/courses/bio104/cellresp.htm]
(2) Antranik [http://antranik.org/cell-respiration-part-1-anaerobic-respiration-glycolysis-and-fermentation/]
- Cellular respiration is an aerobic reaction which requires oxygen. Fermentation does not require oxygen but does release lactic acid which causes our cells and muscles to weaken.
(1) Clc [http://biology.clc.uc.edu/courses/bio104/cellresp.htm]
(2) Antranik [http://antranik.org/cell-respiration-part-1-anaerobic-respiration-glycolysis-and-fermentation/]
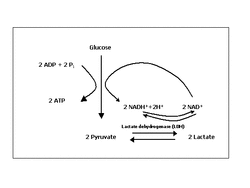
8. Recognize the role of glycolysis in oxidizing glucose to two molecules of pyruvate
- During glycolysis, two ATP molecules are used to break down the glucose into two pyruvate molecules. The net yield of glycolysis is 2 ATP and 2 NADH.
(1) Elm [http://www.elmhurst.edu/~chm/vchembook/601glycolysisrx.html]
(2) Ncbi [http://www.ncbi.nlm.nih.gov/books/NBK22395/]
- During glycolysis, two ATP molecules are used to break down the glucose into two pyruvate molecules. The net yield of glycolysis is 2 ATP and 2 NADH.
(1) Elm [http://www.elmhurst.edu/~chm/vchembook/601glycolysisrx.html]
(2) Ncbi [http://www.ncbi.nlm.nih.gov/books/NBK22395/]

9. Describe the process that brings pyruvate from the cytosol into the mitochondria and introduces it into the citric acid cycle.
- The pyruvate moves from cytosol into the mitochondria by a transport protein. In the matrix of mitochondria, the CO2 is removed by an enzyme and NAD+ convert to NADH. Then it adds coenzyme A to form Acetyl CoA to enter the citric acid cycle.
(1) UCD [http://chemwiki.ucdavis.edu/Biological_Chemistry/Metabolism/Kreb%27s_Cycle]
(2) Shmoop [http://www.shmoop.com/cell-respiration/citric-acid-cycle.html]
- The pyruvate moves from cytosol into the mitochondria by a transport protein. In the matrix of mitochondria, the CO2 is removed by an enzyme and NAD+ convert to NADH. Then it adds coenzyme A to form Acetyl CoA to enter the citric acid cycle.
(1) UCD [http://chemwiki.ucdavis.edu/Biological_Chemistry/Metabolism/Kreb%27s_Cycle]
(2) Shmoop [http://www.shmoop.com/cell-respiration/citric-acid-cycle.html]

10. Explain how the process of chemiosmosis utilizes the electrons from NADH and FADH2 to produce ATP
- The electrons from NADH and FADH2 provided energy for the electron transport chain, and it is also used to power for pumping the H+ proton across the inner membrane. Then the flow of H+ back across the membrane provide energy for ATP synthesis.
(1) Cliffs [http://www.cliffsnotes.com/sciences/biology/biology/cellular-respiration/chemiosmosis]
(2) Ask [http://www.ask.com/question/what-is-chemiosmosis]
- The electrons from NADH and FADH2 provided energy for the electron transport chain, and it is also used to power for pumping the H+ proton across the inner membrane. Then the flow of H+ back across the membrane provide energy for ATP synthesis.
(1) Cliffs [http://www.cliffsnotes.com/sciences/biology/biology/cellular-respiration/chemiosmosis]
(2) Ask [http://www.ask.com/question/what-is-chemiosmosis]

11.Write out the summary equation of photosynthesis including the source and fate of the reactants and products
6CO2+6H2O+Light energy→ C6H12O6+6O2
- Plants absorb water through their roots, and carbon dioxide through their leaves. During photosynthesis, the plants get light energy from the sun to help the water reacts with carbon dioxide to produce sugar and oxygen. The glucose is used for respiration or stored in starch. The oxygen is releases as a by-product of photosynthesis.
(1) Clc [http://biology.clc.uc.edu/courses/bio104/photosyn.htm]
(2) Khan [https://www.khanacademy.org/science/biology/photosynthesis]
6CO2+6H2O+Light energy→ C6H12O6+6O2
- Plants absorb water through their roots, and carbon dioxide through their leaves. During photosynthesis, the plants get light energy from the sun to help the water reacts with carbon dioxide to produce sugar and oxygen. The glucose is used for respiration or stored in starch. The oxygen is releases as a by-product of photosynthesis.
(1) Clc [http://biology.clc.uc.edu/courses/bio104/photosyn.htm]
(2) Khan [https://www.khanacademy.org/science/biology/photosynthesis]
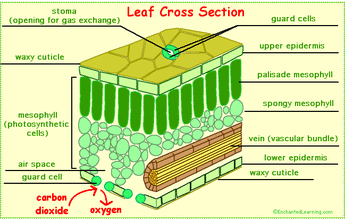
12. Discuss how leaf and chloroplast anatomy relates to photosynthesis
- The chloroplasts are located in the top of the leaf, so it allows for capturing more sun exposure to use as free energy to begin the process of photosynthesis. The stomata is located on the bottom layer of leaf, so gases produced during photosynthesis (H20, CO2, O2) can be regulated by the leaf.
(1) Plp[ http://www.plantphysiol.org/content/155/1/108]
(2)Adap[https://www.adapaonline.org/bbk/tikindex.php?page=Leaf]

13. Describe how photosystems convert solar energy to chemical energy
- Photosystems consist of a light-harvesting complex and a reaction center. In the light-harvesting complex, the chlorophyll will absorb light in the form of photons. Then the energy is transferred to reaction center of the photosystems. At the reaction center, an excited electron of chlorophyll a paired to the primary electron acceptors. This step convert the solar energy to chemical energy.
(1) Ask[http://biology.about.com/od/plantbiology/a/aa050605a.htm]
(2) Uzh[http://www.lightchec.uzh.ch/index.html]
- Photosystems consist of a light-harvesting complex and a reaction center. In the light-harvesting complex, the chlorophyll will absorb light in the form of photons. Then the energy is transferred to reaction center of the photosystems. At the reaction center, an excited electron of chlorophyll a paired to the primary electron acceptors. This step convert the solar energy to chemical energy.
(1) Ask[http://biology.about.com/od/plantbiology/a/aa050605a.htm]
(2) Uzh[http://www.lightchec.uzh.ch/index.html]

14. Explain how linear electron flow in the light reactions results in the formation of ATP, NADPH, and O2
- During the light reaction, electrons flow through the photosystems. Photosystem 2 absorbs the photo of light, and the excited electron boosted to P680 chlorophyll a. An enzyme catalyzing the water to breakdown, so the oxygen atom combines with another oxygen atom to form O2. After the electron passed the primary acceptor, the electron move through the electron transport chain to photosystem 1. As the electron transfers to a low energy level, the exergonic reaction of electron released energy is used to pump protons and create a H+ gradient by chemiosmosis to phosphorylate ADP to ATP. The primary acceptor of photosystem 2 transfer electron to another ETC, then the enzyme NADP+ catalyzes .
(1) Lib [http://library.thinkquest.org/27819/ch4_10.shtml]
(2) Nco[http://www.nature.com/ncomms/2013/130613/ncomms2954/fig_tab/ncomms2954_F1.html]
- During the light reaction, electrons flow through the photosystems. Photosystem 2 absorbs the photo of light, and the excited electron boosted to P680 chlorophyll a. An enzyme catalyzing the water to breakdown, so the oxygen atom combines with another oxygen atom to form O2. After the electron passed the primary acceptor, the electron move through the electron transport chain to photosystem 1. As the electron transfers to a low energy level, the exergonic reaction of electron released energy is used to pump protons and create a H+ gradient by chemiosmosis to phosphorylate ADP to ATP. The primary acceptor of photosystem 2 transfer electron to another ETC, then the enzyme NADP+ catalyzes .
(1) Lib [http://library.thinkquest.org/27819/ch4_10.shtml]
(2) Nco[http://www.nature.com/ncomms/2013/130613/ncomms2954/fig_tab/ncomms2954_F1.html]

15. Illustrate how chemiosmosis generates ATP in the light reactions
- In the light reactions, when the electron transfer through an electron transport chain. This ETC pumps protons to create a H+ gradient that used in chemiosmoses to add phosphate group to bond with ADP to form ATP.
(1) RCN [http://users.rcn.com/jkimball.ma.ultranet/BiologyPages/L/LightReactions.html]
(2) BCS [http://bcs.whfreeman.com/thelifewire/content/chp08/0802002.html]
- In the light reactions, when the electron transfer through an electron transport chain. This ETC pumps protons to create a H+ gradient that used in chemiosmoses to add phosphate group to bond with ADP to form ATP.
(1) RCN [http://users.rcn.com/jkimball.ma.ultranet/BiologyPages/L/LightReactions.html]
(2) BCS [http://bcs.whfreeman.com/thelifewire/content/chp08/0802002.html]

16. Summarize how the Calvin cycle uses the energy molecules of the light reactions to produce G3P
- In the Calvin cycle, each 3-phosphoglycerate get one phosphate group from ATP to form 1,3-bi phosphoglycerate. Then the electrons of NADPH reduces the 1,3-bi phosphoglycerate molecules to glyceraldehyde 3-phosphate(G3P). It cost nine molecules of ATP and sic molecules of NADPH produced by the light reaction.
(1) Umass [http://www.bio.umass.edu/biology/conn.river/calvin.html]
Ccbs [http://student.ccbcmd.edu/~gkaiser/biotutorials/photosyn/fg7.html]
- In the Calvin cycle, each 3-phosphoglycerate get one phosphate group from ATP to form 1,3-bi phosphoglycerate. Then the electrons of NADPH reduces the 1,3-bi phosphoglycerate molecules to glyceraldehyde 3-phosphate(G3P). It cost nine molecules of ATP and sic molecules of NADPH produced by the light reaction.
(1) Umass [http://www.bio.umass.edu/biology/conn.river/calvin.html]
Ccbs [http://student.ccbcmd.edu/~gkaiser/biotutorials/photosyn/fg7.html]
In Unit 3, we learned that cellular work uses ATP by combining exergonic and endergonic reactions. Chemical work can be the synthesis of polymer, transport work can be the pumping of substances cross the membrane, mechanical work can be the muscle contraction. Understand cellular respiration teaches us how cells harness chemical energy and use to generate ATP, which drives most cellular work. Photosynthesis provide energy for the entire living world directly and indirectly because plants get energy from the sun without eating other organisms. On lands, plants perform photosynthesis, and in water photosynthetic organisms are many: multicellular algae, unicellular protist, and cya-no bacteria.
Vocabulary (Chapter 8-10)
1.Metabolism is the totality of an organism’s chemical reactions
2.Induced fit of a substrate brings chemical groups of the active site into positions that enhance their ability to catalyze the reaction
3.Allosteric regulation may either inhibit or stimulate an enzyme’s activity
4.Cooperativity is a form of allosteric regulation that can amplify enzyme activity
5.Obligate anaerobes carry out fermentation or anaerobic respiration and cannot survive in the presence of O2
6.Facultative anaerobes are bacterias can survive using either fermentation or cellular respiration
7.Autotrophs sustain themselves without eating anything derived from other organisms
8.Heterotrophs obtain their organic material from other organisms
9.The electromagnetic spectrum is the entire range of electromagnetic energy, or radiation
10.Action spectrum profiles the relative effectiveness of different wavelengths of radiation in driving a process
1.Metabolism is the totality of an organism’s chemical reactions
2.Induced fit of a substrate brings chemical groups of the active site into positions that enhance their ability to catalyze the reaction
3.Allosteric regulation may either inhibit or stimulate an enzyme’s activity
4.Cooperativity is a form of allosteric regulation that can amplify enzyme activity
5.Obligate anaerobes carry out fermentation or anaerobic respiration and cannot survive in the presence of O2
6.Facultative anaerobes are bacterias can survive using either fermentation or cellular respiration
7.Autotrophs sustain themselves without eating anything derived from other organisms
8.Heterotrophs obtain their organic material from other organisms
9.The electromagnetic spectrum is the entire range of electromagnetic energy, or radiation
10.Action spectrum profiles the relative effectiveness of different wavelengths of radiation in driving a process
