Unit 2 covers carbon composition, the formation of organic compounds, cell structure and functions, and cell membranes. Carbon forms molecules by reducing free energy. The diverse molecules arise from this relate to evolution because different structures means different functions. Many large molecules such as carbohydrates, lipids, proteins and nuclei acids have may different functions (e.g: nucleic acids store hereditary information). All cell functions come from complex system interactions of these molecules. The cell membrane’s lipid bilayer structure comes from minimizing free energy and transportation across the membrane transmits information in larger system interactions.
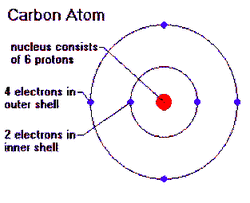
1. Identify the properties of carbon that make it so important
- All organic compounds contain carbon. Carbon has four valence electrons to form 4 covalent bonds. This property help carbon atoms can form diverse and large molecules by bonding to four other atoms. For example, carbohydrates, protein, and fates all contain carbon.
(1) Yzi[http://www.youngzine.org/article/why-all-life-carbon-based]
(2) Co[http://oolong.co.uk/carbon]
- All organic compounds contain carbon. Carbon has four valence electrons to form 4 covalent bonds. This property help carbon atoms can form diverse and large molecules by bonding to four other atoms. For example, carbohydrates, protein, and fates all contain carbon.
(1) Yzi[http://www.youngzine.org/article/why-all-life-carbon-based]
(2) Co[http://oolong.co.uk/carbon]

2. Discuss the role of dehydration reactions in the formation of organic compounds and hydrolysis in the digestion of organic compounds
- In the dehydration reaction, two monomers joined together by removing one water molecule. Then the monomers build up to polymers. In the hydrolysis, the large molecule is split into small molecule by adding a water molecule.
(1) Lone[http://nhscience.lonestar.edu/biol/dehydrat/dehydrat.html]
(2) Tripod[http://thepeaceorg.tripod.com/organic/id8.html]
- In the dehydration reaction, two monomers joined together by removing one water molecule. Then the monomers build up to polymers. In the hydrolysis, the large molecule is split into small molecule by adding a water molecule.
(1) Lone[http://nhscience.lonestar.edu/biol/dehydrat/dehydrat.html]
(2) Tripod[http://thepeaceorg.tripod.com/organic/id8.html]
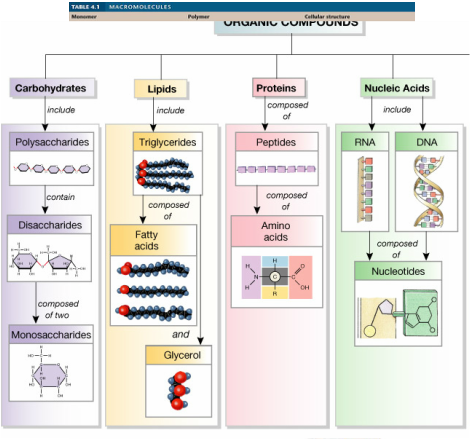
3. Recognize the four biologically important organic compounds (carbohydrates, lipids, proteins, and nucleic acids) by their structural formulas
- Carbohydrates are molecular compounds made of carbon, hydrogen and oxygen. Lipids are made of fatty acids and glycerol. Proteins are polypeptides made of amino acids, which consist of a carboxyl group, an amino group, a hydrogen atom, and a R group. Nucleic acids are made of nucleotides, which consist of a sugar, a nitrogen base, and a phosphate group.
(1)Wiki[http://en.wikibooks.org/wiki/Structural_Biochemistry/Organic_Chemistry/Carbohydrates]
(2) Clic[http://biology.clc.uc.edu/courses/bio104/lipids.htm]
- Carbohydrates are molecular compounds made of carbon, hydrogen and oxygen. Lipids are made of fatty acids and glycerol. Proteins are polypeptides made of amino acids, which consist of a carboxyl group, an amino group, a hydrogen atom, and a R group. Nucleic acids are made of nucleotides, which consist of a sugar, a nitrogen base, and a phosphate group.
(1)Wiki[http://en.wikibooks.org/wiki/Structural_Biochemistry/Organic_Chemistry/Carbohydrates]
(2) Clic[http://biology.clc.uc.edu/courses/bio104/lipids.htm]
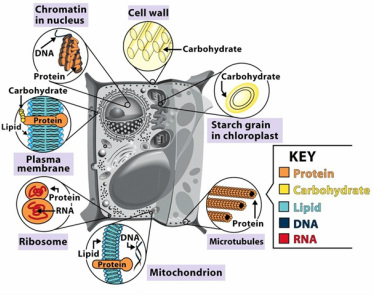
4. Explain the cellular functions of the four groups of organic compounds
- Carbohydrates can fuel cellular respiration and build materials. It can also provide structural support and used as energy storage.
- Lipids are important energy source as energy storage and protect vital organs and insulation.
- Proteins have vary functions. It can be used as enzymes to catalyze chemical reactions, and as transport molecules to transport substances. It also provide structural support and receive signals from outside cell.
- Nucleic acids stores all the hereditary information and transmit genetic materials.
(1)Cliff[http://www.cliffsnotes.com/sciences/biology/biology/the-chemical-basis-of-life/organic-compounds]
(2)Usc[http://www.ucs.mun.ca/~iemerson/lectures1001/coursenotes/Functionsorganicmolecules.html]
- Carbohydrates can fuel cellular respiration and build materials. It can also provide structural support and used as energy storage.
- Lipids are important energy source as energy storage and protect vital organs and insulation.
- Proteins have vary functions. It can be used as enzymes to catalyze chemical reactions, and as transport molecules to transport substances. It also provide structural support and receive signals from outside cell.
- Nucleic acids stores all the hereditary information and transmit genetic materials.
(1)Cliff[http://www.cliffsnotes.com/sciences/biology/biology/the-chemical-basis-of-life/organic-compounds]
(2)Usc[http://www.ucs.mun.ca/~iemerson/lectures1001/coursenotes/Functionsorganicmolecules.html]

5. Specify the four structural levels of proteins and how changes at any level can affect the activity of the protein
- There are four structure levels of proteins: primary structure is its unique sequence of amino acids, secondary structure is segments of polypeptide chains folded in patterns. Tertiary structure is the patterns is the patterns of folding polypeptide chains. Quaternary is the overall structure from the aggregation of subunits. If there is a change in the sequence of the amino acids, it will change the shape and function of the protein. If the secondary and tertiary structure is disrupted, it will cause the protein denaturation
(1) Biotopics[http://www.biotopics.co.uk/JmolApplet/proteinjstructure.html]
(2) Cvmb[http://arbl.cvmbs.colostate.edu/hbooks/genetics/biotech/basics/prostruct.html]
- There are four structure levels of proteins: primary structure is its unique sequence of amino acids, secondary structure is segments of polypeptide chains folded in patterns. Tertiary structure is the patterns is the patterns of folding polypeptide chains. Quaternary is the overall structure from the aggregation of subunits. If there is a change in the sequence of the amino acids, it will change the shape and function of the protein. If the secondary and tertiary structure is disrupted, it will cause the protein denaturation
(1) Biotopics[http://www.biotopics.co.uk/JmolApplet/proteinjstructure.html]
(2) Cvmb[http://arbl.cvmbs.colostate.edu/hbooks/genetics/biotech/basics/prostruct.html]

6. Recall how proteins reach their final shape (conformation), the denaturing impact that heat and pH can have on protein structure, and how these changes may affect the organism
- Proteins consist of amino acids which are polar with positive and negative charge. The change of pH means the change of the amount of H+ atoms. If there is a change in ph, the hydrogen atom with positive charge will attract the negative side of amino acids, so it will cause unstable in the protein structure and denature it. Heat will disrupt the interactions of hydrogen bonds between amino acid. Then the structure will be unstable and denatured. When the protein is denatured, its shape and function will also changed, then the denatured protein will no longer effective.
(1) Elm[http://www.elmhurst.edu/~chm/vchembook/568denaturation.html]
(2) Sciencedaily[http://www.sciencedaily.com/articles/d/denaturation_%28biochemistry%29.htm]
- Proteins consist of amino acids which are polar with positive and negative charge. The change of pH means the change of the amount of H+ atoms. If there is a change in ph, the hydrogen atom with positive charge will attract the negative side of amino acids, so it will cause unstable in the protein structure and denature it. Heat will disrupt the interactions of hydrogen bonds between amino acid. Then the structure will be unstable and denatured. When the protein is denatured, its shape and function will also changed, then the denatured protein will no longer effective.
(1) Elm[http://www.elmhurst.edu/~chm/vchembook/568denaturation.html]
(2) Sciencedaily[http://www.sciencedaily.com/articles/d/denaturation_%28biochemistry%29.htm]

7. List three differences between prokaryotic and eukaryotic cells
- Prokaryotic cells do not have nucleus but eukaryotic cells do.
- Prokaryotic cells do not have many organelles but eukaryotic cells do.
- Prokaryotic cells are unicellular but eukaryotic cells are multicultural.
(1)Diffen[http://www.diffen.com/difference/Eukaryotic_Cell_vs_Prokaryotic_Cell]
(2)Cod[http://www.cod.edu/people/faculty/fancher/prokeuk.htm]
- Prokaryotic cells do not have nucleus but eukaryotic cells do.
- Prokaryotic cells do not have many organelles but eukaryotic cells do.
- Prokaryotic cells are unicellular but eukaryotic cells are multicultural.
(1)Diffen[http://www.diffen.com/difference/Eukaryotic_Cell_vs_Prokaryotic_Cell]
(2)Cod[http://www.cod.edu/people/faculty/fancher/prokeuk.htm]

8. Discuss the structure and function of organelles common to plant and animal cells
- Nucleus contains genetic information
- Mitochondria is for cellular respiration and generate ATP
- Ribosomes make proteins
- Golgi apparatus transport proteins
- Endoplasmic reticulum is a network fro membranes
- Plasma membrane is selective permeable and control the material in or out of cell
- Peroxisome specialized metabolic functions
(1)Think[http://library.thinkquest.org/12413/structures.html]
(2) Junction[http://www.biologyjunction.com/cell_functions.htm]
- Nucleus contains genetic information
- Mitochondria is for cellular respiration and generate ATP
- Ribosomes make proteins
- Golgi apparatus transport proteins
- Endoplasmic reticulum is a network fro membranes
- Plasma membrane is selective permeable and control the material in or out of cell
- Peroxisome specialized metabolic functions
(1)Think[http://library.thinkquest.org/12413/structures.html]
(2) Junction[http://www.biologyjunction.com/cell_functions.htm]
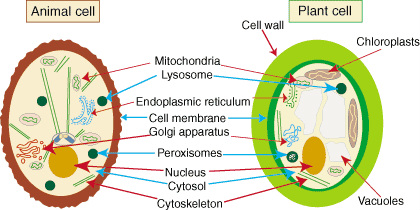
9. Specify the structure and function of organelles found only in plant cells or only in animal cells
Only in plant cells
- Chloroplast is for photosynthesis that convert light energy to chemical energy
- Central Vacuole function as storage, hydrolysis macromolecules
- Cell wall maintains cell shape and protect cell
Only in animal cells
- Lysosomes is digestive organelle to hydrolysis macromolecule
- Centriole is microtubules that pull apart chromosomes during cell division
- Flagella is long micro-tubes used for directed cell movement
- Cilia is hair-like organelles for cell movement
Ss[http://www.slideshare.net/jayak1/02-organelles-in-animal-and-plant-cells]
Bol[http://www.biology-online.org/11/1_plant_cells_vs_animal_cells.htm]
Only in plant cells
- Chloroplast is for photosynthesis that convert light energy to chemical energy
- Central Vacuole function as storage, hydrolysis macromolecules
- Cell wall maintains cell shape and protect cell
Only in animal cells
- Lysosomes is digestive organelle to hydrolysis macromolecule
- Centriole is microtubules that pull apart chromosomes during cell division
- Flagella is long micro-tubes used for directed cell movement
- Cilia is hair-like organelles for cell movement
Ss[http://www.slideshare.net/jayak1/02-organelles-in-animal-and-plant-cells]
Bol[http://www.biology-online.org/11/1_plant_cells_vs_animal_cells.htm]

10. Explain why membranes are selectively permeable
- Cell membranes are selectively permeable that it allows some substances to cross more easily than others, because membranes serves as a barrier that separate the inner cell and the surrounding environment. It needs to control the substances in or out of the cell. It can prevent harmful materials from entering the cell.
(1)Stcc[http://faculty.stcc.edu/AandP/AP/AP1pages/Units1to4/introduction/selectiv.htm]
(2)Phs[ http://www.phschool.com/science/biology_place/biocoach/biomembrane1/permeability.html]
- Cell membranes are selectively permeable that it allows some substances to cross more easily than others, because membranes serves as a barrier that separate the inner cell and the surrounding environment. It needs to control the substances in or out of the cell. It can prevent harmful materials from entering the cell.
(1)Stcc[http://faculty.stcc.edu/AandP/AP/AP1pages/Units1to4/introduction/selectiv.htm]
(2)Phs[ http://www.phschool.com/science/biology_place/biocoach/biomembrane1/permeability.html]

11. Identify the role of phospholipids, proteins, and carbohydrates in membranes
- The phospholipids in the membrane can separate the cell from its liquid environment. Hydrophilic molecules can’t enter the cell but hydrophobic can. Unsaturated hydrocarbon tails of phospholipids kept the molecules from packing together, which enhancing the fluidity of membrane.
- There are two types of proteins in the membrane: integral protein and peripheral proteins. The functions of membrane proteins are transport, enzymatic activity, signal transduction, and cell-cell recognition. For transport, it can form channels or pores to let substances pass through. For example, polar molecules and ions require specific transport proteins.
- Carbohydrates are the short chain of sugars attached to the outside surface of membrane, they interact with surface molecules of other cells for cell-cell recognition.
(1) Ken[http://biology.kenyon.edu/HHMI/Biol113/membranes.htm]
(2) Ncbi[http://www.ncbi.nlm.nih.gov/books/NBK9928/]
- The phospholipids in the membrane can separate the cell from its liquid environment. Hydrophilic molecules can’t enter the cell but hydrophobic can. Unsaturated hydrocarbon tails of phospholipids kept the molecules from packing together, which enhancing the fluidity of membrane.
- There are two types of proteins in the membrane: integral protein and peripheral proteins. The functions of membrane proteins are transport, enzymatic activity, signal transduction, and cell-cell recognition. For transport, it can form channels or pores to let substances pass through. For example, polar molecules and ions require specific transport proteins.
- Carbohydrates are the short chain of sugars attached to the outside surface of membrane, they interact with surface molecules of other cells for cell-cell recognition.
(1) Ken[http://biology.kenyon.edu/HHMI/Biol113/membranes.htm]
(2) Ncbi[http://www.ncbi.nlm.nih.gov/books/NBK9928/]

12. Predict how water will move if a cell is placed in an isotonic, hypertonic, or hypotonic solution and the effect of different environments on the organism
- In an isotonic solution, there is no water movement across the plasma membrane. Water flows across the membrane at same rates in both directions; the cell is stable in the isotonic environment.
- In an hypertonic solution, the cell will lose water to its surrounding where has more solutes, because water will move the high concentration of solutes. The cell lose water to the environment, so the cell will shrivel or die.
- In an hypotonic solution, the cell will gain much water from its surrounding where has less solutes, because the cell has more solutes that water will come in the cell faster than it leaves. In a hypotonic environment, the cell will sell or burst.
(1)Phd[http://www.phschool.com/science/biology_place/biocoach/biomembrane1/solutions.html]
(2) Ss[http://www.slideshare.net/dewisivasamy/the-effects-of-hypotonic-hypertonic-and-isotonic]
- In an isotonic solution, there is no water movement across the plasma membrane. Water flows across the membrane at same rates in both directions; the cell is stable in the isotonic environment.
- In an hypertonic solution, the cell will lose water to its surrounding where has more solutes, because water will move the high concentration of solutes. The cell lose water to the environment, so the cell will shrivel or die.
- In an hypotonic solution, the cell will gain much water from its surrounding where has less solutes, because the cell has more solutes that water will come in the cell faster than it leaves. In a hypotonic environment, the cell will sell or burst.
(1)Phd[http://www.phschool.com/science/biology_place/biocoach/biomembrane1/solutions.html]
(2) Ss[http://www.slideshare.net/dewisivasamy/the-effects-of-hypotonic-hypertonic-and-isotonic]

13. Illustrate how electrochemical gradients are formed and function in cells
- Electrochemical gradients are formed by two forces: chemical force and electrical force. The chemical force is the difference in ion concentration. The electrical force is the difference in charges across the membrane. These two forces drives the diffusion of ions across the membrane.
(1)Boundless[https://www.boundless.com/biology/structure-and-function-of-plasma-membranes/active-transport/electrochemical-gradient/]
(2)Arbl[http://arbl.cvmbs.colostate.edu/hbooks/cmb/cells/pmemb/transport_active.html]
- Electrochemical gradients are formed by two forces: chemical force and electrical force. The chemical force is the difference in ion concentration. The electrical force is the difference in charges across the membrane. These two forces drives the diffusion of ions across the membrane.
(1)Boundless[https://www.boundless.com/biology/structure-and-function-of-plasma-membranes/active-transport/electrochemical-gradient/]
(2)Arbl[http://arbl.cvmbs.colostate.edu/hbooks/cmb/cells/pmemb/transport_active.html]
In Unit 2, we learned an asymmetric carbon can create enantiomers with different structure and function: ibuprofen treats pain and information, and albuterol treats asthma. Understanding organic compounds can be used to innovate new biological technology such as chitin from cicada can be used to make a degradable surgical threads. Muscle contraction is caused by cytoskeleton (actin filaments). Cell membranes have membrane proteins with many functions: such as transport, enzymatic activity, signal transduction, cell-cell recognition and etc.
Vocabulary (Chapter 4-7)
1.Hydrocarbons are organic molecules consisting of only carbon and hydrogen
2.Vitalism is the idea that organic compounds arise only in organisms, was disproved when chemists synthesized these compounds
3.Mechanism is the view that all natural phenomena are governed by physical and chemical laws
4.Isomers are compounds with the same molecular formula but different structures and properties
5. Antiparallel is an arrangementv in the DNA double helix that the two backbones run in opposite 5′→ 3′ directions from each other
6.Plasmodesmata are channels that perforate plant cell walls
7.Cell fractionation takes cells apart and separates the major organelles from one another
8.Tonicity is the ability of a surrounding solution to cause a cell to gain or lose water
9.Osmoregulation is the control of solute concentrations and water balance, is a necessary adaptation for life in such environments
10.The fluid mosaic model states that a membrane is a fluid structure with a “mosaic” of various proteins embedded in it
1.Hydrocarbons are organic molecules consisting of only carbon and hydrogen
2.Vitalism is the idea that organic compounds arise only in organisms, was disproved when chemists synthesized these compounds
3.Mechanism is the view that all natural phenomena are governed by physical and chemical laws
4.Isomers are compounds with the same molecular formula but different structures and properties
5. Antiparallel is an arrangementv in the DNA double helix that the two backbones run in opposite 5′→ 3′ directions from each other
6.Plasmodesmata are channels that perforate plant cell walls
7.Cell fractionation takes cells apart and separates the major organelles from one another
8.Tonicity is the ability of a surrounding solution to cause a cell to gain or lose water
9.Osmoregulation is the control of solute concentrations and water balance, is a necessary adaptation for life in such environments
10.The fluid mosaic model states that a membrane is a fluid structure with a “mosaic” of various proteins embedded in it
