Unit 1 addresses all the big themes: evolution, free energy, information, and system interaction. Biological hierarchy and organism interaction and structure and function give rise to complex system interactions. DNA and feedback mechanisms transfer information. Chemical bonds are made and broken to minimize free energy to make chemical reactions which are essential for system interaction. The polarity of water also cause the minimization of free energy to cause important system interaction on life.
1. Demonstrate how evolution is a change in gene frequency in a population over time
- Gene is the units of inheritance that transmit information from parents and offsprings. Genes are contain within DNA, which store information. DNA produces protein and RNA. Protein and RNA define an organism’s phenotype. Certain phenotype has an advantage in natural selection while other phenotype die out. So, the population’s genetic composition changes. Evolution is defined as a change in a population’s genetic composition from generation to generation.
(1) Scitable [http://www.nature.com/scitable/definition/evolution-78]
(2) Science daily [http://www.sciencedaily.com/articles/e/evolution.htm]
- Gene is the units of inheritance that transmit information from parents and offsprings. Genes are contain within DNA, which store information. DNA produces protein and RNA. Protein and RNA define an organism’s phenotype. Certain phenotype has an advantage in natural selection while other phenotype die out. So, the population’s genetic composition changes. Evolution is defined as a change in a population’s genetic composition from generation to generation.
(1) Scitable [http://www.nature.com/scitable/definition/evolution-78]
(2) Science daily [http://www.sciencedaily.com/articles/e/evolution.htm]
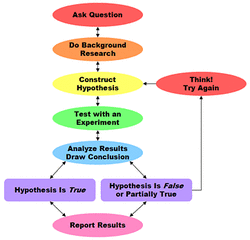
2. Summarize the process of science
- The process of science begins with an observation and then asking a question about the observation. Then a few hypotheses are formed, which explain the observation. An experiment is designed to test the hypotheses from which you record data. Then you get the final result which will tell you if your hypothesis is right or wrong after analyzing the data. Finally, you make a conclusion to summarize your results.
(1) Science Buddies [http://www.sciencebuddies.org/science-fair-projects/project_scientific_method.shtml]
(2) Understanding Science [http://undsci.berkeley.edu/article/0_0_0/howscienceworks_02]
- The process of science begins with an observation and then asking a question about the observation. Then a few hypotheses are formed, which explain the observation. An experiment is designed to test the hypotheses from which you record data. Then you get the final result which will tell you if your hypothesis is right or wrong after analyzing the data. Finally, you make a conclusion to summarize your results.
(1) Science Buddies [http://www.sciencebuddies.org/science-fair-projects/project_scientific_method.shtml]
(2) Understanding Science [http://undsci.berkeley.edu/article/0_0_0/howscienceworks_02]
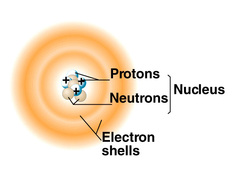
3. Identify the three subatomic particles and their significance
- An atom, the smallest unit of matter, is composed of subatomic particles. Three major subatomic particles are protons, neutrons, and electrons. Protons and neutrons are found in the nucleus, and electrons are in the electron shells around the nucleus. Protons have positive charges and determine the element because each element has a unique number of protons. Neutrons have no charge and determine isotopes because their numbers vary in the same element. Electrons have negative charges and determine chemical properties.
(1) Science Daily [http://www.sciencedaily.com/articles/s/subatomic_particle.htm]
(2) NDT [http://www.ndt-ed.org/EducationResources/HighSchool/Radiography/subatomicparticles.htm]
- An atom, the smallest unit of matter, is composed of subatomic particles. Three major subatomic particles are protons, neutrons, and electrons. Protons and neutrons are found in the nucleus, and electrons are in the electron shells around the nucleus. Protons have positive charges and determine the element because each element has a unique number of protons. Neutrons have no charge and determine isotopes because their numbers vary in the same element. Electrons have negative charges and determine chemical properties.
(1) Science Daily [http://www.sciencedaily.com/articles/s/subatomic_particle.htm]
(2) NDT [http://www.ndt-ed.org/EducationResources/HighSchool/Radiography/subatomicparticles.htm]
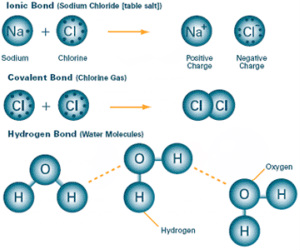
4. Explain the types of chemical bonds, how they form, and their relative strengths
- Chemical bonds are interactions between the valance electrons of different atoms. The attractions held the atoms together to form molecules.
- Covalent bonds form when two atoms share the valence elections. In non-polar covalent bonds, the electrons are shared equally between atoms. In polar covalent bonds, the atom with higher electronegativity, a tendency to attract electrons, will get more electrons.
- Ionic bonds form when two charged atoms called ions will attract each other because of opposite charges. The iron with positive charge is a cation. The one with negative charge is an anion.
- Hydrogen bonds occurs when a hydrogen atom covalently bonded to an electronegativity atom.
- Van der Waals Interactions occurs when two atoms are very close to each other. It is the weakest chemical bonds.
- The relative strength of the bonds from strongest to weakest are covalent bonds, ionic bonds, hydrogen bonds, and Van der Waals interactions.
(1) NDT [http://www.ndt-ed.org/EducationResources/CommunityCollege/Materials/Structure/bonds.htm]
(2) NCBI [http://www.ncbi.nlm.nih.gov/books/NBK22567/]
- Chemical bonds are interactions between the valance electrons of different atoms. The attractions held the atoms together to form molecules.
- Covalent bonds form when two atoms share the valence elections. In non-polar covalent bonds, the electrons are shared equally between atoms. In polar covalent bonds, the atom with higher electronegativity, a tendency to attract electrons, will get more electrons.
- Ionic bonds form when two charged atoms called ions will attract each other because of opposite charges. The iron with positive charge is a cation. The one with negative charge is an anion.
- Hydrogen bonds occurs when a hydrogen atom covalently bonded to an electronegativity atom.
- Van der Waals Interactions occurs when two atoms are very close to each other. It is the weakest chemical bonds.
- The relative strength of the bonds from strongest to weakest are covalent bonds, ionic bonds, hydrogen bonds, and Van der Waals interactions.
(1) NDT [http://www.ndt-ed.org/EducationResources/CommunityCollege/Materials/Structure/bonds.htm]
(2) NCBI [http://www.ncbi.nlm.nih.gov/books/NBK22567/]
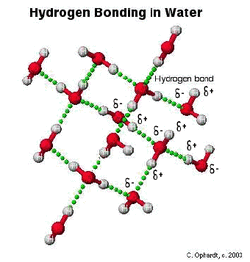
5. Explain the importance of hydrogen bonding to the properties of water?
- The slightly positive hydrogen of one water molecule will attach to the slightly negative oxygen of a nearby water molecule. These two molecules are held together by the hydrogen bonds to form a polar molecules, which means the two ends of this molecule have opposite charges. Cohesion and adhesion occur because of hydrogen bonding. Water has a high specific heat. The hydrogen bonds absorb heat to break down and release heat to form. It requires high energy to change the temperature of water. Ice is less dense than liquid water because hydrogen bonding pushes the molecules apart in a lattice. Water is also an important solvent because it binds to hydrophilic molecules.
(1) How stuff works [http://science.howstuffworks.com/environmental/earth/geophysics/h2o7.htm]
(2) Inforplease [http://www.infoplease.com/cig/biology/water.html]
- The slightly positive hydrogen of one water molecule will attach to the slightly negative oxygen of a nearby water molecule. These two molecules are held together by the hydrogen bonds to form a polar molecules, which means the two ends of this molecule have opposite charges. Cohesion and adhesion occur because of hydrogen bonding. Water has a high specific heat. The hydrogen bonds absorb heat to break down and release heat to form. It requires high energy to change the temperature of water. Ice is less dense than liquid water because hydrogen bonding pushes the molecules apart in a lattice. Water is also an important solvent because it binds to hydrophilic molecules.
(1) How stuff works [http://science.howstuffworks.com/environmental/earth/geophysics/h2o7.htm]
(2) Inforplease [http://www.infoplease.com/cig/biology/water.html]
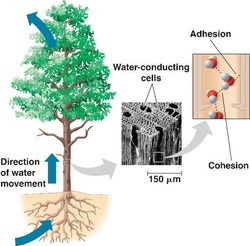
6. Describe four unique properties of water, and how each contributes to life on Earth
- The property of cohesion contribute to the transportation of water in plants. The water molecules cling to each other help transport from the roots to the leaves. The adhesion of water to cell walls help resist the downward gravity.
- Water has a high specific heat. This property of high specific heat help water has a steady temperature to support variety life when the weather changes. It also helps with the evaporation. When the surface temperature is high, water will absorb the heat to cool the surface.
- Ice floating occurs because water as solid is less dense than as liquid. Ice floats in water. This help forms ice on the tops of water and leave a habitat underneath.
- Water is an important solvent, the substances that something can dissolved in. Hydrophilic substances are water-soluble ( e.g. proteins and sugars), so water can carry those materials around in the organisms. Hydrophobic substances such as oils are non-polar and do not dissolve in water. Lipid bilayer and protein folding requires hydrophilic and hydrophobic interactions.
(1) Dummies [http://www.dummies.com/how-to/content/the-unusual-properties-of-water-molecules.html]
(2) Shmoop [http://www.shmoop.com/biomolecules/properties-water.html]
- The property of cohesion contribute to the transportation of water in plants. The water molecules cling to each other help transport from the roots to the leaves. The adhesion of water to cell walls help resist the downward gravity.
- Water has a high specific heat. This property of high specific heat help water has a steady temperature to support variety life when the weather changes. It also helps with the evaporation. When the surface temperature is high, water will absorb the heat to cool the surface.
- Ice floating occurs because water as solid is less dense than as liquid. Ice floats in water. This help forms ice on the tops of water and leave a habitat underneath.
- Water is an important solvent, the substances that something can dissolved in. Hydrophilic substances are water-soluble ( e.g. proteins and sugars), so water can carry those materials around in the organisms. Hydrophobic substances such as oils are non-polar and do not dissolve in water. Lipid bilayer and protein folding requires hydrophilic and hydrophobic interactions.
(1) Dummies [http://www.dummies.com/how-to/content/the-unusual-properties-of-water-molecules.html]
(2) Shmoop [http://www.shmoop.com/biomolecules/properties-water.html]

7. Interpret the pH scale
- The pH scale measure s the relative acidity and alkalinity of substances. It ranges from 0 to 14. The pH of 7 is neutral. The pH is less than 7 is acidic, which means have an excess of H+ ions. The pH is more than 7 is basic, which means have an excess of OH- ions.
(1) Elmhurts [http://www.elmhurst.edu/~chm/vchembook/184ph.html]
(2)EPA [ http://www.sciencebuddies.org/science-fair-projects/project_ideas/Chem_AcidsBasespHScale.shtml]
- The pH scale measure s the relative acidity and alkalinity of substances. It ranges from 0 to 14. The pH of 7 is neutral. The pH is less than 7 is acidic, which means have an excess of H+ ions. The pH is more than 7 is basic, which means have an excess of OH- ions.
(1) Elmhurts [http://www.elmhurst.edu/~chm/vchembook/184ph.html]
(2)EPA [ http://www.sciencebuddies.org/science-fair-projects/project_ideas/Chem_AcidsBasespHScale.shtml]
8. Predict how changes in pH can alter biological systems
- In the gastric system, if you have too much acid in your stomach, it will damage stomach lining. If the pH of blood is too high, it will damage blood cells. You would lose bone density because the minerals are going to the blood as buffer from the bone.
(1) Ehow [http://www.ehow.com/about_6165622_can-affected-change-ph-levels_.html]
(2) Boundless [https://www.boundless.com/biology/the-chemical-foundation-of-life/wate/phbuffers-acids and-bases/]
- In the gastric system, if you have too much acid in your stomach, it will damage stomach lining. If the pH of blood is too high, it will damage blood cells. You would lose bone density because the minerals are going to the blood as buffer from the bone.
(1) Ehow [http://www.ehow.com/about_6165622_can-affected-change-ph-levels_.html]
(2) Boundless [https://www.boundless.com/biology/the-chemical-foundation-of-life/wate/phbuffers-acids and-bases/]

9. Discuss the importance of buffers in biological systems
- Buffers are substances that minimize changes in pH. For example, the carbonic acid moderates pH changes in blood plasma and the ocean.
(1) Wisc [ http://scifun.chem.wisc.edu/chemweek/biobuff/biobuffers.html]
(2) Sparknotes [http://www.sparknotes.com/chemistry/acidsbases/buffers/section1.html]
- Buffers are substances that minimize changes in pH. For example, the carbonic acid moderates pH changes in blood plasma and the ocean.
(1) Wisc [ http://scifun.chem.wisc.edu/chemweek/biobuff/biobuffers.html]
(2) Sparknotes [http://www.sparknotes.com/chemistry/acidsbases/buffers/section1.html]
In Unit 1, We learned the fundamental elements of life in this unit. The evolution leads to biodiversity; for example, different species of finches with different diets arise from natural selection because of differences in available food in different environment. Chemical bounds form compounds such as table salt, which allows us to get essential but uneatable elements like sodium and chloride. The high specific heat from the polarity of water affects climate near the large body of water. Proper function in the body and coral reefs are sensitive to pH of blood and sea water, and carbonic acid is used as a buffer to stabilize the pH in both.
Vocabulary (Chapter 1-3)
1. Evolution is the process of change that has transformed life on Earth
2.Emergent properties result from the arrangement and interaction of parts within a system
3.Genome of an organism is its entire set of genetic instructions
4.Element is a substance that cannot be broken down to other substances by chemical reactions
5.Isotopes are two atoms of an element that differ in number of neutrons
6.Chemical equilibrium is reached when the forward and reverse reaction rates are equal
7.Specific heat of a substance is the amount of heat that must be absorbed or lost for 1 g of that
8.Buffers are substances that minimize changes in concentrations of H+ and OH– in a solution
9.Solution is a liquid that is a homogeneous mixture of substances
10.Solvent is the dissolving agent of a solution
1. Evolution is the process of change that has transformed life on Earth
2.Emergent properties result from the arrangement and interaction of parts within a system
3.Genome of an organism is its entire set of genetic instructions
4.Element is a substance that cannot be broken down to other substances by chemical reactions
5.Isotopes are two atoms of an element that differ in number of neutrons
6.Chemical equilibrium is reached when the forward and reverse reaction rates are equal
7.Specific heat of a substance is the amount of heat that must be absorbed or lost for 1 g of that
8.Buffers are substances that minimize changes in concentrations of H+ and OH– in a solution
9.Solution is a liquid that is a homogeneous mixture of substances
10.Solvent is the dissolving agent of a solution
